Polydextrose – from seed to Eureba
Product development • With an ability to provide the same volume and consistency as sugar – with 75 per cent fewer calories and almost no effect on blood sugar levels – polydextrose is like a miracle ingredient that makes the bun light and juicy. The tasteless dietary fibre has barely noticeable sweetness, but many other properties in common with sugar (and fat). Polydextrose is produced from the naturally occurring substances glucose, sorbitol and citric acid. In practice, production starts with wheat or other starch-rich crops. But how is it done? We will tell.
Consumers demand less sugar to keep calories in check and reduce the effect on blood sugar levels. But it is rarely as simple as adding less if any sugar at all. We can easily compensate the sweetness with a high-intensity sweetener, for example, steviol glycosides. The big challenge is to maintain the perfect mouthfeel. No one wants an ice cream that doesn’t melt the right way in the mouth or an energy bar that crunch between teeth in a wrong way between. Then polydextrose can be the solution. It is an ingredient that provides softness and juiciness in the same way as fat and provides volume, texture and mouthfeel like sugar.
A successful recipe
In the 1960s, Hans Rennhard and his research team at Pfizer experimented with natural ingredients to produce low-calorie substitutes for sugar, fat, flour and starch. The experimentation gave results in 1965. They mixed 89 per cent glucose, 10 per cent sorbitol and 1 per cent citric acid, heated the mixture and got polydextrose.
Although polydextrose is produced from natural ingredients, the substance itself is not found in nature, and could, therefore, be patented. Pfizer did so in 1973. Then, years of studies followed on the food’s technical properties and how safe it is for humans. It turned out to have suitable technical properties and not only be harmless but even great for people. It was approved as dietary fibre in the United States in 1982. The EU followed 1995.
Glucose
The main ingredient in the production of polydextrose is glucose. The monosaccharide is abundant in fruits and berries, but for food production, glucose is made from starch from natural sources such as corn, wheat, potatoes and cassava.
Manufacturing starts with dissolving starch in water and adding acid or enzymes (or both) and heating everything up. This makes the starch, which consists of long chains of glucose molecules, to break up into increasingly shorter and shorter chains. A chain reaction if you wish. The treatment is called hydrolysis and the result is glucose syrup – a caloric bomb with sky-high glycaemic index (GI).

Sorbitol
The other ingredient in the production of polydextrose is sorbitol. It is a sugar alcohol found naturally in plums, pears, peaches, apples and a host of different fruits and berries. The substance is also found in rowan berries, and this is where it was first discovered. However, industrially, sorbitol is produced from glucose. You can read how it’s done in a separate article on sorbitol.
Citric acid
The third and last ingredient in the production of polydextrose is citric acid. As the name suggests, it is found in citrus fruits, but also in a variety of other fruits and berries.
Citric acid can be extracted from, among other things, unripe lemons and other citrus fruits. That is how it usually was produced from the end of the 19th century and well into the 20th century. But as early as 1917, the American food chemist James Currie discovered that the fungus Aspergillus niger – commonly known as black mould – is a capable producer of citric acid. Practically all citric acid sold today is produced by black mould.
The fact that black mould is used to produce food is a hot topic in some circles on the internet. But there is no cause for concern. There are non-toxic strains of black mould used in the production of citric acid. Thus, it is not the same strains that create harmful mould in damp houses.
Citric acid production begins with the hydrolysis of starch to produce glucose syrups with which the fungus than are fed. In return, they produce citric acid, which is extracted in several steps which includes purification, evaporation, crystallization and drying.

Preparation of polydextrose
We now have all the ingredients necessary to make polydextrose: glucose, sorbitol and citric acid. Note that in practice, all three are made from starch, which comes from wheat, corn, potatoes, cassava or another starch-rich crop.
Polydextrose is prepared by mixing 89 parts of glucose, 10 parts of sorbitol and 1 part of citric acid and heating the mixture till it becomes liquid. Then the citric acid acts as a catalyst and makes glucose and sorbitol to react with each other to form shorter, and longer glucose chains, which in turn are attached to each other and form branched molecules of glucose parts.
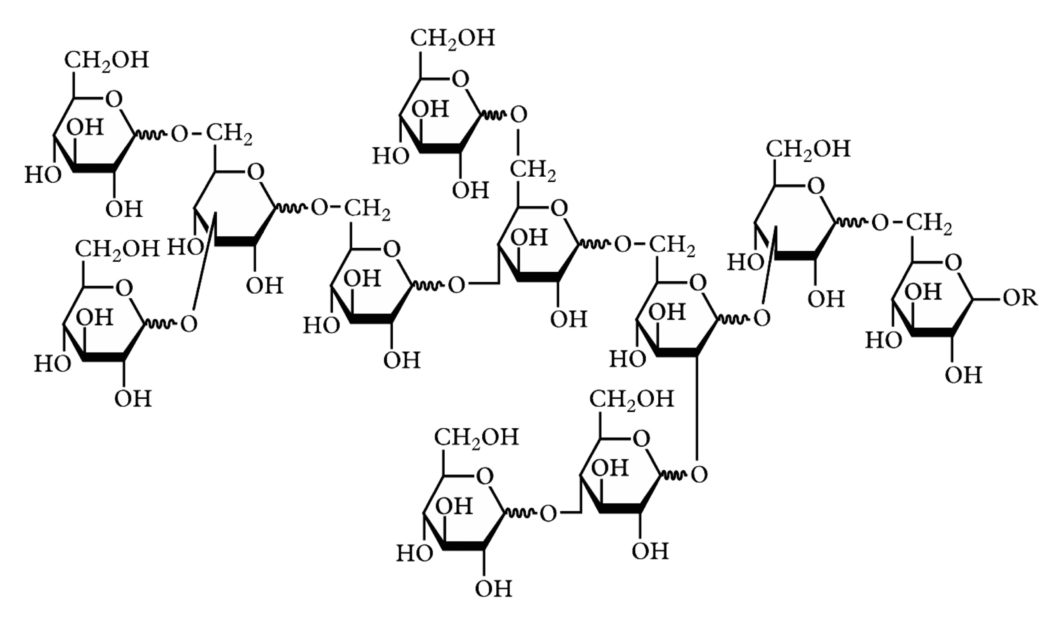
It is these branched molecules of glucose that are polydextrose. On average, they have twelve glucose molecules, but the number can vary from three to over one hundred. That is why they are called polydextrose; poly means many in classical Greek, and dextrose is another name for glucose.
But all glucose and sorbitol are not converted. And the citric acid, which was just a catalyst, remains. Also, some levoglucose, which is a naturally occurring organic substance, has been formed.
Polydextrose is dietary fibre
So what makes polydextrose a fibre? The answer lies in how the glucose molecules are attached to each other.
When a carbon atom in one glucose molecule and a carbon atom in another glucose molecule hold the same oxygen atom in their hands, they become linked. The two carbon atoms and the oxygen atom is called a glycoside bond. These are denoted by α or β followed by two numbers, for example, α-(1→4) and β-(1→6). The numbers tell which carbon atoms hold the oxygen atom in their hands, and the Greek letter says whether the carbon atoms are turned in different or the same direction. (You will find a more detailed description with illustrations in the article on dextrin.)
A polydextrose molecule has a jumble of glycoside bonds: α- and β-(1→2), (1→3), (1,→4) and (1→6). There are most of α-(1→6) and β-(1→6).
Although they are called almost the same thing, our bodies handle them very differently. Our digestive system quickly breaks down α-(1→4) bonds, which are the glycoside linkage in starch and common sugars, but struggle with α- and β-(1→6) bonds. If a glucose chain has more than one α- or β-(1→6) binding, the body does not have time to break it down before it reaches the large intestine. Such carbohydrates are called dietary fibres.
Since α- and β-(1→6) are common in polydextrose, most of it will pass the stomach and small intestine before without being touched. This, together with the fact that polydextrose has a favourable physiological effect as scientific demonstrated, makes polydextrose approved as dietary fibre.
Better on the stomach
Polydextrose is better on the stomach than sugar alcohols. Studies show that you can eat up to 50 grams of polydextrose at one time, and up to 90 grams during a day without stomach problems. The limit values are well above the 25 to 35 grams of fibre that an adult should eat per day (and fewer than half come up to). Therefore, polydextrose may be used without restriction in most foods, but not more than necessary.
Several features in the same ingredient

Polydextrose has many useful properties that make it a popular ingredient in everything from drinks, ice cream and pastries to dressings and jams. With neutral taste, it’s a chameleon that fits in many contexts.
The gastric-friendly dietary fibre, made from natural ingredients, is a good starting point for creating a sweetened fibre. Since polydextrose is not sweet in itself, it must be supplemented with a sweetener such as steviol glycosides and/or sugar alcohols. Precisely what to complement polydextrose and in what proportions vary from application to application.
We can help you
Don’t hesitate to contact us if you want help finding the right ingredients and proportions to replace sugar with sweetened fibre. Chances are that we already have a ready-made solution for you. If not, we can help you develop one.
Please, share this article if you liked it.
[et_social_share]





